
ICODOS has demonstrated its technology at a pilot e-methanol plant.Credit: A. Bramsiepe/KIT
ICODOS: Fuelling industry with waste gases
Methanol produced from captured carbon dioxide could yield hydrogen to power the green economy sustainably.
Mission
To create methanol for the chemical and shipping industries, using captured carbon dioxide and hydrogen produced with renewable energy at a volume and cost that will be attractive to industry.
Main achievement
The company has demonstrated its method in a pilot plant in Karlsruhe, Germany, and won a €2-million (US$2.1-million) grant from the European Union science funding mechanism Horizon Europe to build a demonstration plant 15 times larger than the pilot plant, with a capacity of 150 tonnes of e-methanol per year.
Technology
ICODOS combines carbon capture and methanol synthesis into a single process. First, the company pumps industrial waste gas into a high-pressure solvent made of water and methanol, which absorbs CO2 from the gas.
The CO2 is then removed from the liquid and mixed with hydrogen, which is derived from water electrolysis powered by renewable energy. High pressures and temperatures then convert that mixture into a hydrogen and methanol mix that is free of CO2. Some of that mix goes back into the solvent to continue the process, while the rest is distilled into methanol and water. The design reduces the amount of energy required to capture carbon by 30% compared with existing technologies.
ICODOS also says that its process can reduce the cost of producing e-methanol — named because it uses renewable energy in its synthesis — by 17%. The company also makes the plant more efficient by using machine learning to create an automated control system that adapts to variations in the availability of solar and wind power. The automation reduces labour and maintenance costs by 75%.
Biggest challenge
Designing and building plants to capture carbon from industrial sources and produce e-methanol requires time and money. So far, e-methanol production has yet to be implemented at large scales. The current supply is less than 1 million tonnes per year, but demand for e-methanol is projected to be 17 million tonnes by 2030.
Potential impact
The chemical and shipping industries account for about 10% of global greenhouse-gas emissions. Every year, the chemical industry uses about 120 million tonnes of methanol produced from fossil-fuel sources as a feedstock. Shipping burns about 210 million tonnes of fuel per year. Replacing fossil methanol with e-methanol could reduce the carbon emissions that contribute to climate change by as much as one gigatonne per year by 2050, according to ICODOS projections.
Judge’s comment
“ICODOS have clearly identified and understand the problem facing the industry and how their proposed solution could contribute to its resolution,” says Sue Sundstrom, a mentor for early-stage technology companies and executive director of Sundstrom Innovation in Clevedon, UK.
LigniLabs: Environmentally friendly crop protection
Protecting grape vines from destructive and hard-to-kill fungi.

LigniLabs have come up with a way to target pesticides for grape vines.Credit: Holger Leue/Getty
Mission
To encapsulate pesticides for wine grapes and other crops, thereby reducing the amount of pesticide required, targeting it for its intended purpose, and reducing the release of microplastics into the environment.
Main achievement
The company has created ESCApe, a targeted pesticide and delivery mechanism that combats a previously untreatable disease that destroys grapevines, known as esca.
Technology
The key component is an encapsulant made from lignin, an organic polymer that is one of the two major components of plant tissue. The encapsulant forms a shell around materials, such as pesticides.
In the case of ESCApe, it encloses a pesticide designed to attack the fungi that cause esca. These hide inside the trunk of the grapevine and are therefore inaccessible to sprayed fungicides. The lignin microcarriers are 200–700 nanometres in diameter and are injected into the sap through a hole drilled in the vine. They become active only when an enzyme produced by the fungi degrades the lignin shell and releases the pesticide.
Biggest challenge
Designing the microcarriers is complicated. They must be the right size and have the right properties to flow through the sap without aggregating. There also needs to be a mechanism to release their contents, but they can’t leak. It has also been difficult to scale-up production from microlitres to litres per week. The company will need to study the safety of its products thoroughly to gain regulatory approval, because they’re intended for use with crops that are meant for human consumption.
Potential impact
Offering treatments that are tailored to specific plant diseases or pests reduces the amount of pesticide needed and reduces unwanted effects on other crops. LigniLabs’ first product could help the wine industry, which loses around €1 billion worldwide each year to esca.
Using a wood-like polymer could reduce plastic pollution, with pesticides and fertilizers responsible for an estimated 10,000 tonnes of microplastic per year in Europe alone. LigniLabs hopes to tackle that problem further by expanding to other types of pesticide, such as for bark beetles that damage forests. Another goal is to replace plastic encapsulants used in a wide variety of products, including flame retardants, paints and materials for 3D printing.
Judge’s comment
“It’s an exciting application of nature-derived biomaterials,” says Hyunwoo Yuk, a mechanical engineer and co-founder of the bioadhesive company SanaHeal — winner of The Spinoff Prize 2023. It’s particularly timely, Yuk adds, given the growing concern about microplastics and ‘forever chemicals’ in the environment.
Lumetallix: Getting the lead out
Detecting this toxic metal is the first step in protecting children from it.

Lumetallix’s lead-detection spray glows green to reveal lead in the soil.Credit: AMOLF
Mission
To create an affordable and reliable test to detect lead in various materials. The test is intended especially for use in low- and middle-income countries with high levels of childhood lead toxicity.
Main achievement
A lead detection kit was made commercially available in August 2023.
Technology
Lead combines with various halogen atoms, including fluorine, chlorine and iodine, to form lead halide perovskites — crystals widely used in making light-emitting diodes and solar cells. Lumetallix created a reagent that contains precursors to perovskites and additives to encourage their reaction with lead. When sprayed onto lead, the reagent forms perovskites that glow under ultraviolet light, creating an easily visible signature.
Laboratory tests show that the method can detect lead at levels as low as 1 nanogram per square millimetre, which is 1,000 times more sensitive than the conventional method of reacting the lead with sodium rhodizonate. Moreover, whereas the sodium rhodizonate test returns false positives up to 78% of the time, the company’s perovskite-generating spray produced no false positives during testing. The approach is also significantly less expensive than laboratory tests that use mass spectrometers or X-ray fluorescence analysers.
The company has demonstrated its technology on paint, plastics, alloys, ceramics, solder, dust, batteries and soil.
Biggest challenge
Lumetallix hopes organizations such as the US Environmental Protection Agency will accept its test as a new lead-testing standard.
Potential impact
An estimated 800 million children have lead poisoning, which can affect physical and mental development. Lead is thought to cost the world $6 trillion annually in health care and lost productivity. Lumetallix hopes its test will allow pregnant people to test their homes for lead contamination to protect their newborns. The company also intends for the technology to be used to detect lead in drinking water and in blood, and even improve forensic tests for gunshot residue.
Judge’s comment
“It’s a very exciting company aiming to provide more affordable and easy-to-use solutions for the critical problem of lead contamination. It was one of my favourite companies during the review process,” says Yuk. “It’s a really creative use of perovskite. Lead contamination is a serious issue in many developing countries, so Lumetallix’s easy and affordable solution will be able to bring big social impacts.”
Organoids derived from people’s tumours reveal which drugs are likely to work.
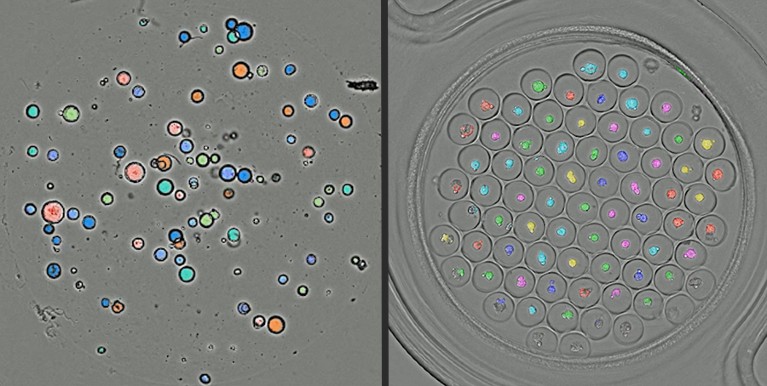
Orbits Oncology use artificial intelligence to monitor ‘organoids’ in domes (left) and well plates (right).Credit: C. Deben et al. Cell. Oncol. 46, 299–314 (2023).
Mission
To replace animal models and cell lines for cancer-drug testing with digital surrogates, based on personal data and tests conducted on cells grown from individuals with cancer.
Main achievement
A pilot study compared conventional high-throughput screening with Orbits Oncology’s digital-surrogate technique, screening 1,280 compounds on 6,144 wells containing tissue samples. The company’s analysis produced useful data in less than 24 hours, compared with more than 19 days for standard techniques.
Technology
The company works with patient-derived organoids (PDOs) — cancer models made directly from people’s tumours that retain some biological features specific to that person. Orbits Oncology applied machine-learning techniques to develop an algorithm that uses microscope imagery to track the PDOs. This captures their response to drugs over time, without disturbing the models.
The company can then combine the results from PDO testing with personal information, such as ethnicity, previous therapies and genomic profile, and ask an artificial-intelligence model to predict the best treatments.
Biggest challenge
PDOs are a new, and not thoroughly understood, technology. Academic labs often lack the expensive equipment that will allow them to go beyond rudimentary analysis, and only about 10–15% of biopharmaceutical companies use PDOs for drug development. As a result, the usefulness of Orbits Oncology’s technology as a supplement to safety and toxicity studies in preclinical development has yet to be established.
Potential impact
The cost of pharmaceuticals remains high because so many drugs (around 90%, by some estimates) fail in clinical trials. These dead ends cost roughly $60 billion in research and development, annually. If Orbits Oncology does a better job of representing a person with cancer than current models do, it could filter out more candidates at the preclinical stage, saving a lot of that money.
Judge’s comment
The company’s plan to develop the technology is well thought out, says Sundstrom. However, she notes that it does not include immunotherapy — a substantial part of the cancer-treatment market.
Fluorescent molecules put a spotlight on a type of ubiquitin ligase that plays a part in diseases such as cancer.
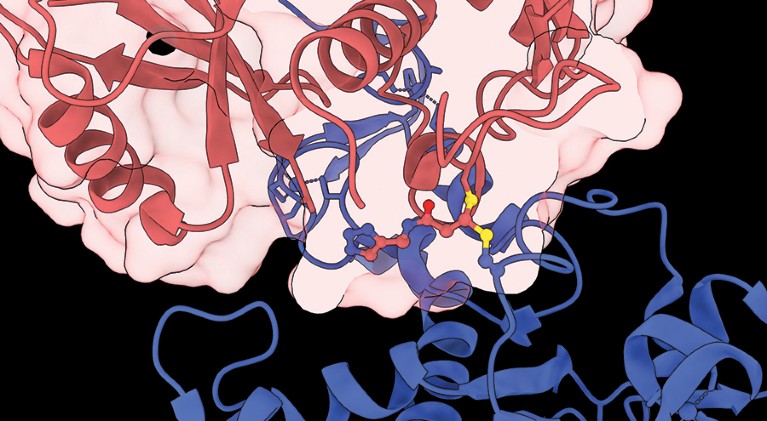
A ubiquitin E3 ligase (blue) labelled by a protein-based sensor of E3 activity (red).Credit: Outrun Therapeutics
Mission
To develop drugs that target E3 ubiquitin ligases, which affect the stability of proteins and are involved in diseases such as some cancers and neurodegenerative disorders.
Main achievement
Outrun Therapeutics has identified a single target in a cancer and is using its technology to develop a drug candidate for that target. The company hopes to identify a candidate drug to take into clinical trials by the end of the year.
Technology
An activity-based probe (ABP) is a small molecule that fluoresces when it binds to and reacts with the active site of an enzyme. Although ABPs have proved useful in unlocking the secrets of proteins, none existed for E3 ubiquitin ligases until Outrun Therapeutics’ founders developed one.
E3 ligases, of which around 700 are encoded by the human genome, play an important part in the regulation of proteins, and, therefore, can be an important factor in cancer, autoimmune diseases, neurodegeneration and infectious disease. Armed with probes to detect E3, the company is identifying targets for drugs that could stabilize the associated proteins and therefore fight the diseases.
Biggest challenge
The ubiquitin system is complicated, and few drugs have been approved that target E3 ligases. Outrun Therapeutics has so far identified two possible targets, and is developing one of them; there is a long road from there to successful clinical trials and regulatory approval.
Potential impact
E3 ligases have been implicated in numerous diseases, including cancers such as colorectal cancer, head and neck cancers, breast cancer and ovarian cancer. Developing drugs that affect these ligases opens up a therapeutic area and could provide new treatments for millions of people, including those with autoimmune disorders, neurodegenerative diseases and infections.
Judge’s comment
“ABPs are a truly elegant chemical biology solution to inhibitor identification,” says Sundstrom. E3 ligase inhibitors could prove to be an easier avenue for a drug programme than other methods under development, she adds.
A fluorescent dye helps to assess the condition and points to the best treatment.
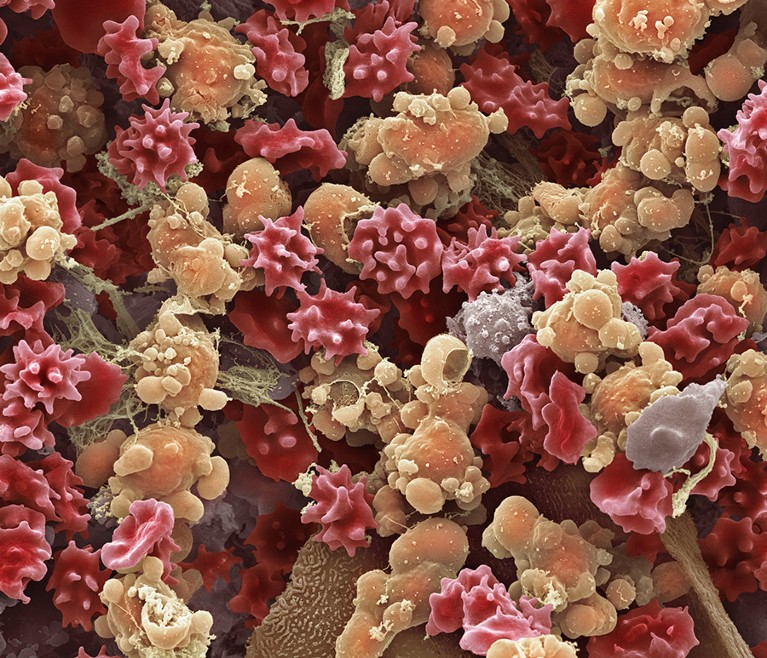
Epithelial and red and white blood cells in a urine sample taken from someone with a urinary tract infection.Credit: Steve Gschmeissner/SPL
Mission
To create a rapid point-of-care diagnostic test for urinary tract infections that can also identify the bacterial species responsible for the infection and whether it is resistant to common antibiotics — all within an hour.
Main achievement
The company has built and tested prototypes of each component of its system. The next step is to combine them into a single unit.
Technology
Testing for urinary tract infections currently involves culturing samples on a laboratory plate for more than 48 hours, and counting the number of colony-forming units of bacteria using flow cytometry. The PhenUtest approach replaces this with a microscope-based assay that uses a live-staining fluorescent dye.
The test also uses one growth medium designed to promote growth and another dosed with an antibiotic to impede growth. If there is no antibiotic resistance, the two media show as much as a fourfold difference in the number of cells within 30 minutes.
Biggest challenge
Clinical samples might vary in ways not seen in lab-stored samples. If that turns out to be the case, PhenUtest might find it difficult to demonstrate a full prototype in a real-world setting by the end of the year. The company still needs to validate its test clinically to satisfy regulatory agencies.
Potential impact
Hundreds of millions of people around the world have urinary tract infections each year. Because of the long wait for test results, physicians prescribe antibiotics solely on the basis of their best guess about what the infectious agent is. Treatments fail in more than 12% of people — and the incorrect and unnecessary use of antibiotics breeds antimicrobial resistance. PhenUtest’s technology could change this.
Furthermore, collecting information from the tests in different parts of the world could help to map antimicrobial resistance, which might act as a warning system for future bacterial pandemics.
Judge’s comment
Combining the rapidity of the test with the ability to detect resistance are important innovations, says Kiana Aran, a bioengineer at the University of California, San Diego. “This could help in potentially minimizing the growth of antimicrobial resistance, addressing major health-care costs and treatment efficiency globally,” she says.
Seedsight: Ferreting out food fraud
Artificial-intelligence technology could help to ensure the safety and nutritional value of the grains that sustain society.

Seedsight is creating a digital repository of the ‘signatures’ of seeds and grain.Credit: Bloomberg Creative/Getty
Mission
To improve the efficiency and safety of the food-processing industry by authenticating the provenance of grains to prevent food fraud, and by measuring levels of nutrients and contaminants.
Main achievement
Food fraud involves adding or substituting mislabelled, inferior and sometimes dangerous ingredients in food products and costs between $10 billion and $40 billion annually, according to the US Food and Drug Administration. A pilot project with a European cereal producer identified wheat’s country of origin with 90% accuracy. It also predicted how much flour could be extracted from the grain with 75% accuracy. Two more pilot studies are scheduled for this year, one in Nigeria and one in Ghana.
Technology
A laser beam is aimed at tiny particles of grain; analysis of the scattered light yields information about the grain’s biomolecular and physical properties, which helps to identify characteristics such as a grain’s geographic origin, flour-extraction capacity, protein level, water content and nanoplastic concentration.
Artificial intelligence (AI) models can use those properties to find a signature for particular crops, and predict which grains will have the best yield for a given product. Future goals include identifying allergens in grain during the factory process and measuring the doses of compounds that might be converted to carcinogens during production. The company hopes to expand to a wider variety of grains, as well as fruits and vegetables.
Biggest challenge
The AI models require large amounts of data from several sources around the world.
Potential impact
Seedsight hopes to reduce food-production costs by giving producers more information, in less time, to help them to select which farm’s harvest to use for each product. Seedsight says it can reduce the time it takes producers to select and purchase the harvest that best suits their needs, from two or three months to one week.
The company also says its technology lowers costs by 8%, by letting producers select grain with fewer impurities, and increases productivity by 20%. By lowering costs and improving the quality of crops selected, it could help to address high hunger rates in low-income countries, for instance by finding the best maize (corn) for making baby food in Africa.
Judge’s comment
“The integration of photonics and AI to create a detailed ‘fingerprint’ of grain particles is innovative,” says Emily Mackay, a data scientist at Siemens Energy in Cambridge, UK. However, she notes that Seedsight has yet to complete trials intended to demonstrate the benefits of its technology. The company also still needs to develop its commercialization plan, and work out what regulatory requirements it might need to meet, she adds.
