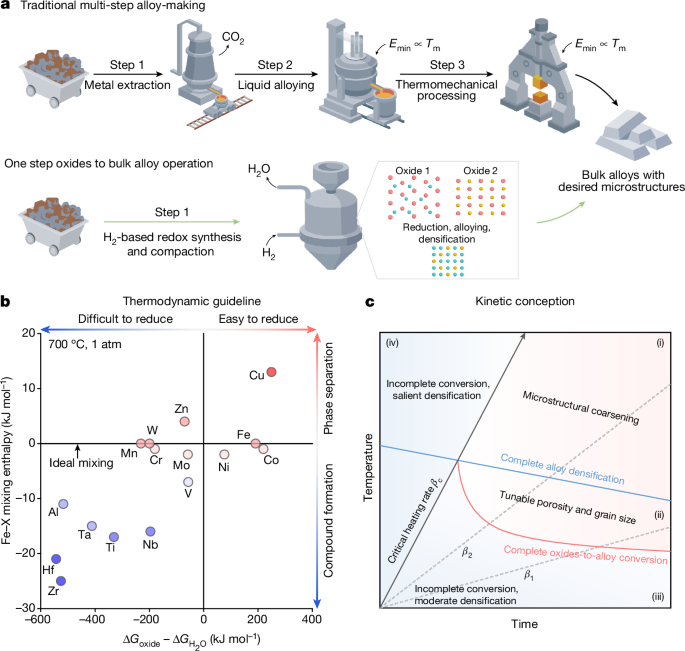
Using H2-based redox reactions, our ‘one step oxides to bulk alloy’ operation (Fig. 1a) is aimed to reform the millennia-old multi-step alloy-making process (Fig. 1a, top) in three aspects: first, eliminating CO2 emission during fossil reductant-based metal extraction; second, reducing the energy cost of liquid processing15,16 that scales with melting temperatures; and third, exploiting the diffusion processes involved directly for compaction. The a priori feasibility of our sustainable alloy synthesis route is governed by the thermodynamic nature of the traditionally separated process steps that we merge here: metal extraction from oxides, atomic-scale mixing amongst the alloying elements and bulk material compaction by diffusion. (Fig. 1a, bottom). Our approach is based on a general thermodynamic design treasure map (Fig. 1b), using the two most important physical parameters involved: solid-state reducibility of oxides in H2, as quantified by \(\Delta {G}_{{\rm{oxide}}}-\Delta {G}_{{{\rm{H}}}_{2}{\rm{O}}}\); and alloying capability, as quantified by the mixing enthalpy between substances (we exemplify Fe–X binary systems in Fig. 1b). Elements in the first and the fourth quadrants (Fe, Ni, Co and Cu) are those that can be fully reduced from their oxides at the solid state by H2, and the closer they locate to the ideal mixing line signifies the more preferential substitutional alloying capability with Fe (Fig. 1b). The thermodynamic validity of our design treasure map well aligns with both historical attempts on alloyed powders17 or nano-composite18 fabrication and the more recent literature on H2-based direct oxide reduction19,20,21.
a, Schematic comparison between the traditional multi-step alloy-making process and the proposed sustainable ‘one step oxides to bulk alloy’ operation. b, Thermodynamically informed design treasure map. For simplicity, here the reducibility (\(\Delta {G}_{{\rm{oxide}}}-\Delta {G}_{{{\rm{H}}}_{2}{\rm{O}}}\)) is considered for the oxides with the highest valence states at 700 °C under 1 atm. Thermodynamic data for constructing this diagram have been collected from the literature2,41 and the SGTE database42. c, Kinetic conception outlining the two main competing factors in achieving bulk alloys with defined microstructures from oxides, related in part to the present demonstrator Fe–Ni alloy class. The physical rationale of such a proposition lies in the difference between the oxide reduction temperature and the temperature at which complete densification is achieved (typically when T/Tm ≈ 0.75, where Tm is the bulk melting point35; see also Extended Data Fig. 1) in the corresponding metallic phase. The critical heating rate, βc, indicates the scenario in which complete oxides-to-alloy conversion and complete densification are simultaneously achieved. β1 and β2 sketch two heating rates slower than βc as a guide to the eye.
For synthesizing not just powders or nanoparticles, but bulk alloys ready for applications, a secondary consideration promptly emerges: sufficient densification and reproducible bulk properties must be attained, which is governed by the kinetics of the underlying mass transport and microstructure formation mechanisms. This design rationale is essential as conventional multi-step alloy-making always requires a third step to reheat the as-cast materials for thermomechanical processing, endowing them with the desired microstructure–property combinations (Fig. 1a, top). Although a quantitatively precise kinetic design guideline relies on the targeted alloy system and product properties, a general conception could still be made, considering the overall interplay between oxides-to-alloy conversion and densification (Fig. 1c). In a theoretical framework encompassing temperature, time and conversion rate, these two phenomena divide the kinetic processing space into four regions, in which a critical heating rate is also involved: our ‘one step oxides to bulk alloy’ operation may be possible only in regions (i) and (ii) in which oxides-to-alloy conversion finishes before complete densification, and further temperature increase in region (i) leads only to salient microstructural coarsening. Regions (iii) and (iv) conversely suggest an incomplete oxides-to-alloy conversion, with moderate or noticeable densification depending on the temperature, respectively. Bearing these semi-quantitative thermodynamic–kinetic guidelines in mind, we next practise our ‘one step oxides to bulk alloy’ concept, aiming to synthesize bulk green Fe–Ni invar alloys as a demonstration. This endeavour is motivated by the substantial environmental costs2,14 associated with fabricating this class of attractive ferrous materials using the conventional extraction–alloying–thermomechanical processing three-step alloy-making procedure.
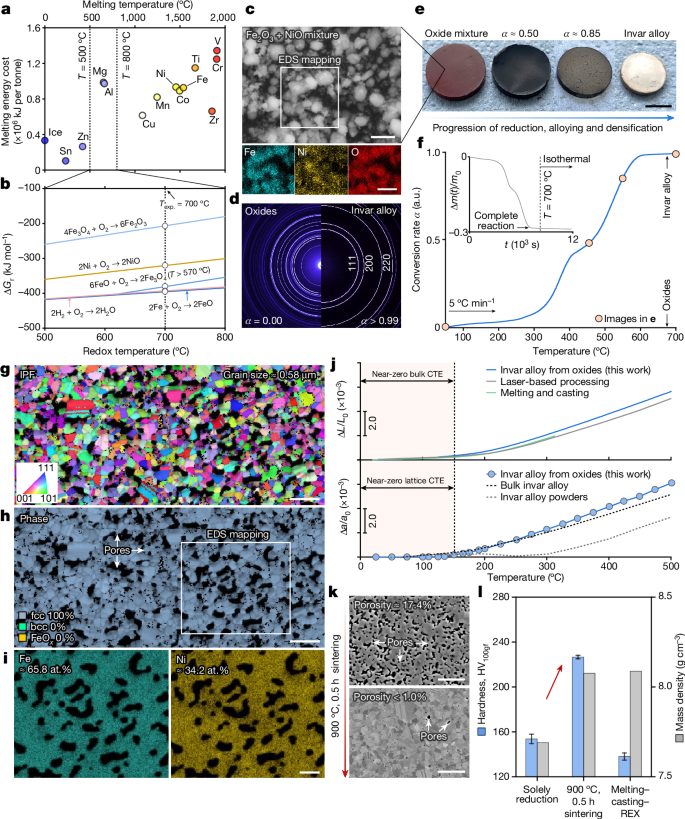
Following the thermodynamic design treasure map (Fig. 1b), we first assess all the possible redox reactions in more quantitative depth. As seen in the calculated Ellingham–Richardson diagram, oxides of Fe and Ni with different valence states all locate above the 2H2 + O2 → 2H2O reaction beyond about 600 °C, suggesting the reducibility of these oxides by H2, and thereby, the formation of metallic Fe and Ni far below their melting points (Fig. 2a,b and Extended Data Fig. 1). To achieve an invar alloy, sufficiently homogeneous mixing between Fe and Ni is indispensable, and the extensive single-phase face-centred cubic (fcc) phase field in the Fe–Ni binary system alleviates such a concern: infinite solubility is present for alloys with more than 20 atomic percent (at.%) Ni above 600 °C. We next mixed Fe2O3 and NiO powders using low-energy ball milling (Fig. 2c,d, left), targeting the Fe–Ni ratio in the invar alloy and compacted them into pellets (Fig. 2e). With this precursor material, we mimic a naturally blended ore with all gangue oxides removed, motivating a proof-of-concept exploration. Secondary electron imaging and energy dispersive X-ray spectroscopy (EDS) confirm the homogeneous mixing of the two oxide powders (Fig. 2c) without discernible mechanical alloying.
a, Minimum melting energy cost as a function of the melting point for common species. The estimation is conducted by adding the enthalpy change of heating up a certain substance from ambient temperature to its melting point and the enthalpy of fusion, that is, \({\Delta E}_{\min }={\int }_{{25}^{^\circ }{\rm{C}}}^{{T}_{{\rm{m}}}}{c}_{{\rm{p}}}\,{\rm{d}}T+{\Delta H}_{{\rm{f}}}\). Only the enthalpy of fusion is considered for ice. Thermophysical parameters for these estimations are acquired from the literature41. b, Predicted Ellingham–Richardson diagram for the oxides of Fe (Fe2O3, Fe3O4 and FeO) and Ni (NiO) under 1 atm (SGTE database42 and refs. 2,41). c, Secondary electron micrograph of the Fe2O3 + NiO powder mixture and the corresponding EDS maps. d, Two-dimensional SXRD diffractograms of the as-compacted oxide pellet (left) and the synthesized invar alloy (right). e, Macroscopic morphological evolution at different conversion rates. f, TGA curve showing the reduction kinetics. Inset: the instantaneous mass loss as a function of time. g, IPF map of the as-synthesized alloy (Σ3 annealing twin boundaries are excluded). h, The corresponding phase map. i, EDS mapping of Fe and Ni. j, Bulk thermal expansion (top, measured using dilatometer) and lattice thermal expansion (bottom, measured using in situ SXRD) results. The bulk and the lattice thermal expansion data for the invar alloys fabricated using different methods are reproduced from the literature25,26,29,30. k, Examples of microstructure tunability. Details of the microstructure after pressure-free sintering are provided in Extended Data Fig. 2. i, Vickers hardness and bulk mass density comparison among the green invar alloys synthesized from oxides and the one fabricated using the conventional melting–casting–recrystallization (REX) method. The recrystallization conditions for the invar alloy processed through melting–casting were also chosen as 900 °C, 0.5 h (around 70% cold-rolling thickness reduction), whose grain size is about 50 μm. a.u., arbitrary units; CTE, coefficient of thermal expansion. Scale bars, 1 μm (c, top and bottom, i); 5 mm (e); 2 μm (g,h); 5 μm (k).
Following the kinetic conception (Fig. 1c), we adopted a moderate heating rate of 5 °C min−1. When heated up in a H2 atmosphere, the pellet undergoes noticeable mass loss accompanied by volumetric shrinkage and colour change (Fig. 2e,f), underpinning the activation of redox reactions. Three stages can be discerned in the thermogravimetric analysis (TGA) (Fig. 2f): (1) reduction initiates at around 290 °C, with a linear increase in conversion rate until an inflection point at about 400 °C, where the reaction is momentarily impeded; (2) the conversion rate resumes to show a linear dependence on temperature in the 450–580 °C range; and (3) an asymptotic trend is present as the conversion rate approaches 0.95 until complete reduction. The presence of distinctive stages in the TGA curve implies the involvement of several redox and alloying micro-events22,23, as rationalized later. At complete reduction, the pellet exhibits a silvery surface, in distinctive contrast with the red-hematite appearance of its as-compacted counterpart (Fig. 2e). Synchrotron X-ray diffraction (SXRD) measurement further validates the single fcc phase constitution of the fully reduced pellet (Fig. 1d, right), in which no remaining oxide phase is detected. The lattice constant of this fcc phase is 3.60 Å. Even considering the uncertainties involved in various measurement techniques, such a lattice constant value agrees well with the literature data for Fe–Ni invar alloys24,25,26 (about 3.60 Å); however, it is notably larger than that of pure fcc-Ni24,27,28 (about 3.52 Å). This distinction evidences substitutional alloying between Fe and Ni during the solid-state redox synthesis, staying consistent with our theoretical anticipation. With this, we prove that the traditionally separate steps of metal extraction and mixing can be merged in a single operation under suitable thermodynamic–kinetic boundary conditions (Fig. 1a).
To assess the ‘oxides to bulk invar alloy’ proposition, we characterize the microstructure and examine the thermal expansion property of the synthesized alloy. Figure 2g shows the electron backscatter diffraction (EBSD) inverse pole figure of the as-synthesized alloy, revealing an equiaxed grain morphology with a fine average grain size of approximately 0.58 μm. Despite the sub-micro-scale porosity owing to the redox-catalysed mass loss and incomplete densification, the microstructure exhibits a single fcc phase constitution (Fig. 2h) without detectable body-centred cubic (bcc) or residual oxide phase at the spatial resolution limit of EBSD (around 50 nm). EDS maps also verify the grain-level uniform distribution of Fe and Ni (Fig. 2i), the contents of which closely follow the conceived values of an invar alloy (Fe–34.8 at.% Ni). The thermal expansion property of the synthesized alloy is next examined using both dilatometry and in situ SXRD. A discernible near-zero coefficient of thermal expansion region is present in the 25–150 °C range for both the bulk and the lattice thermal expansion responses (Fig. 2j). This invar property aligns well with the literature data25,26,29,30 for invar alloys fabricated through the conventional melting–casting–thermomechanical processing routes and even the state-of-the-art laser-based processing methods, again validating our one-step sustainable bulk alloy synthesis approach (Fig. 1a).
The microstructure reported here is mainly aimed to showcase the validity of the ‘one step oxides to bulk alloy’ synthesis concept (Fig. 1), yet, much more diverse microstructure–property combinations can be realized by different kinds of integrated reduction, compaction and microstructure design treatments, implied by our kinetic conception (Fig. 1c). As a simple example, we present in Fig. 2k the possibility of achieving a fine-grain fully densified invar alloy by adding a 0.5 h pressure-free sintering step at 900 °C (correspond to region (i) in Fig. 1c). The average porosity drops from about 17.4% to less than 1%, whereas the average grain size is maintained as about 1.15 μm (Extended Data Fig. 2) and almost all the intercrystalline pores are annihilated, resulting in a bulk mass density nearly identical to that of the invar alloy fabricated through the conventional melting–casting–recrystallization route (Fig. 2l). Because of the salient grain size refinement benefited from the oxide reduction–compaction operation at moderate temperatures, our fully densified invar alloy exhibits a Vickers hardness under 100 g force of 226.6 ± 1.6 HV100gf (Fig. 2l), far exceeding that of the coarse-grained invar alloy obtained from the conventional processing route (138.0 ± 3.2 HV100gf). The foregoing analyses unambiguously substantiate the proposed sustainable alloy synthesis concept (Fig. 1a, bottom) that near-optimized microstructure–bulk property combinations are attained from oxides fully at the solid state. The successful synthesis of the bulk Fe–Ni alloy further reflects the essence of three core physical phenomena at play, as implied by the thermodynamic guideline and the kinetic conception: carbon-free oxide reduction, substitutional alloying and densification for microstructure–property design, altogether motivating mechanistic explorations, as shown next, starting from the phase constitution evolution during reduction.
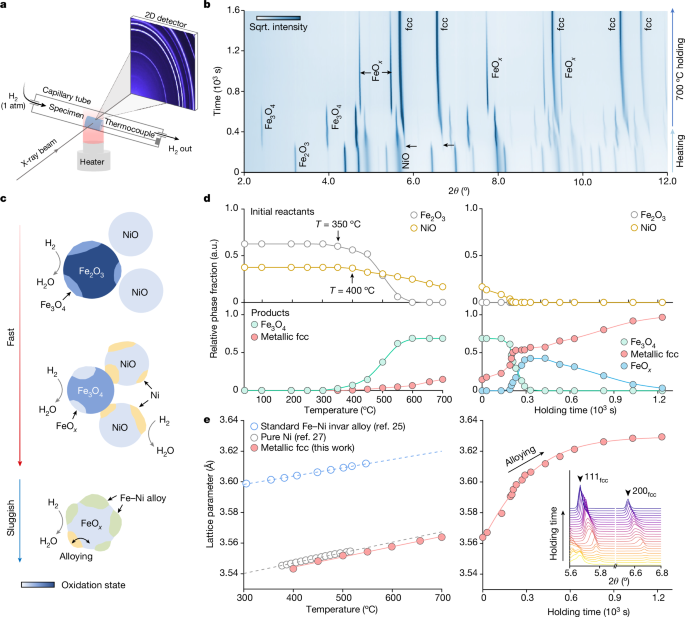
Resorting to the conversion rate curve measured by TGA (Fig. 2f) and the predicted Ellingham–Richardson diagram (Fig. 2b), reduction of the Fe2O3 + NiO mixed oxide should involve several steps, each dictated by the thermodynamics of the redox reaction of the individual oxide. Model-free assessments using the iso-conversional principle31,32 also validate the pronounced dependence of the effective activation energy (Eα) on the local conversion rate (α), indicating the presence of multiple reaction micro-events (Extended Data Figs. 3 and 4). To consolidate this mechanistic proposition, we next performed in situ SXRD measurements (Fig. 3a), characterizing the phase constitution change over time (Supplementary Video 1). The progression of the reaction is shown in Fig. 3b and the schematics in Fig. 3c. The stepwise nature of the reduction process is evident (Fig. 3b), as seen from the quantified relative phase fraction change (Fig. 3d). The first reduction step is Fe2O3 → Fe3O4, which initiates at around 350 °C, leading to the continuous increase of the Fe3O4 phase fraction to about 0.68 at 600 °C (Fig. 3c, top). The reduction of NiO occurs at a slightly higher temperature of around 400 °C, whose fraction monotonically decreases as the reaction advances. The metallic fcc phase emerges from 400 °C (later proved as pure Ni) onwards and its fraction continuously increases to approximately 0.15 as the heating process terminates at 700 °C. On isothermal holding (Fig. 3c, middle, and 3d, right), the NiO phase completely diminishes, associated with the increase of the metallic fcc phase fraction. The Fe3O4 phase fraction remains momentarily constant, followed by the onset of the Fe3O4 → FeOx reaction, leading to a peak FeOx phase fraction of about 0.43. For the rest of the isothermal holding period, the metallic fcc phase fraction keeps increasing and vice versa for the FeOx phase fraction (Fig. 3c, bottom), which accounts for the most sluggish step.
a, Schematic of the experimental set-up and the sample condition. The actual configuration of the experimental instruments is provided in Extended Data Fig. 5. b, Two-dimensional (2D) phase evolution map plotted as a function of time showing the oxide reduction pathway. c, Schematic of the multi-step reduction mechanisms. The colour scale applied in all the oxide phases quantitatively indicates the oxidation state (that is, relative oxygen content). d, Relative phase fraction evolution determined through Rietveld refinement. Note that because of the mass loss during the redox reaction (that is, phase fraction of H2O is unmeasurable by SXRD), datum points in the left and right panels cannot be used to back-derive absolute phase fraction with respect to the reactants. In addition, because of the substantial reaction boundary condition differences between in situ SXRD and TGA, directly correlating these microscopic phase fraction evolution processes with the global conversion rate measurements may not be possible. e, Lattice constant change of the metallic fcc phase observed in the present experiment. Literature data for the pure Ni (ref. 27) and the standard Fe–Ni invar alloy25 are also included in the left panel as references. Inset: the peak shift of 111 and 200 peaks in the metallic fcc phase during 700 °C isothermal holding.
We next focus on solid-state substitutional alloying, the central mechanism in synthesizing the invar alloy. Our rationalization is grounded in the compositionally dependent lattice constants of fcc-structured Fe–Ni alloys. Comparing the lattice constant of the metallic FCC phase obtained here and those of pure Ni (ref. 27) and a standard invar alloy25 (Fig. 3e, left), it is evidenced that pure Ni is the initial metallic phase reduced from the oxide mixture (Fig. 3c, middle). The lattice constant of the metallic fcc phase increases linearly in the 400–700 °C temperature range, consistently aligning with the literature report27 for pure Ni. During isothermal holding (Fig. 3e, right), however, a power-function-like increase in the lattice constant of this phase is present, which eventually plateaus at the lattice constant value of the standard invar alloy. This trend unequivocally reflects the progression of substitutional alloying, which necessitates Fe dissolution into the pure Ni-phase formed earlier and coincides with the prolonged reduction of the FeOx phase.
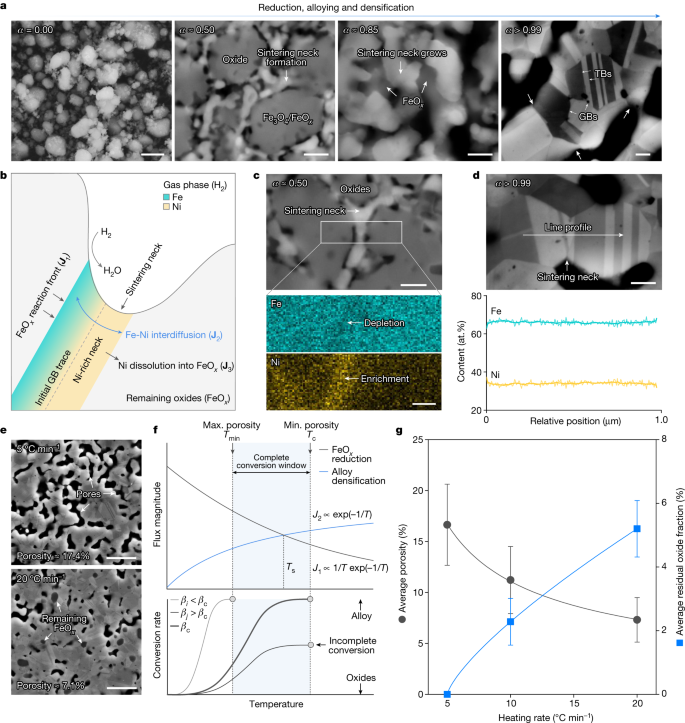
Blending the foregoing thermodynamically oriented insights, we finally explore the governing kinetic phenomena that contribute to the successful invar alloy synthesis and microstructure design directly from the oxides. We suggest that the most prominent rate-limiting process lies in the interplay between interdiffusion-facilitated densification and the FeOx reduction. Pronounced densification occurs during the synthesis, as seen from the considerable volumetric shrinkage of the pellet (Fig. 2e), which follows our kinetic conception (Fig. 1c). As oxide reduction inherently involves volumetric shrinkage33, we present in Extended Data Fig. 6 theoretical calculations, showing that more than 30% of the total volumetric shrinkage is ascribed to sintering-driven densification. Zooming in from macro to micro, we show in Fig. 4a, that the development of the sintering necks corresponds to the material state in Fig. 2e. At a global conversion rate of about 0.5, the formation of metallic interparticle necks initiates and they notably grow as the redox reaction advances to a global conversion rate of about 0.85. Evident densification concurrently operates and the initial necks merge, bringing about multiple grain and annealing twin boundaries at the complete reduction stage. According to the multi-step reaction mechanisms shown earlier, when all the Fe2O3 is reduced to FeOx and NiO to pure Ni, the global conversion rate is about 0.52, implying a direct kinetic overlap among further FeOx reduction, densification and microstructure design.
a, Observations of sintering neck development at different synthesis stages (corresponds to Fig. 2f). b, Schematic of the critical mass transport process. c, EDS analyses of Fe and Ni distribution across a neck at a global conversion rate of around 0.5. d, EDS line profile of Fe and Ni distribution across a neck at a complete reduction. e, Representative secondary electron micrographs of two specimens synthesized using slow and fast heating rates. f, Schematic of the temperature dependency of the two main competing fluxes. Here, J and β denote the flux magnitude of individual mass transport mechanism and the heating rate, respectively. Assuming a constant concentration gradient, the magnitude of the interdiffusion flux facilitating densification, scales with temperature following the Arrhenius law43,44, that is, J2 ∝ exp(−1/T). The flux magnitude for FeOx reduction, takes the form J1 ∝ exp(−Ea/T)[1 − exp(−ΔGr/T)]), as suggested by transition state theory44,45,46, where Ea and ΔGr are the activation energy and the thermodynamic driving force, respectively. The reduction of FeOx in H2 gas exhibits the smallest thermodynamic driving force (ΔGr ≈ 0, with notable backward reaction38,39,47; see also the Ellingham–Richardson diagram in Fig. 2b), allowing us to linearize its temperature dependency to J1 ∝ 1/T exp(−1/T). g, Trade-off between porosity and residual oxide content observed in the specimens obtained using different heating rates. Here the error bars represent the standard deviations. Scale bars, 1 μm (a, for α ≈ 0.00); 200 nm (a, for α ≈ 0.50, α ≈ 0.85 and α > 0.99); 150 nm (c); 200 nm (d); 2 μm (e).
Following these experimental observations, and the earlier overall kinetic conception presented in Fig. 1c, a mechanistic sketch is proposed to detail the governing kinetic processes (Fig. 4b). Two competing mechanisms are considered, the reduction of FeOx by H2 (flux J1) and densification facilitated by Fe–Ni interdiffusion (flux J2). Here we conceive an Ni-rich metallic interparticle neck at the initial state, as supported by the leading NiO → Ni reaction step evidenced by in situ SXRD (Fig. 3) and EDS measurements in Fig. 4c. The role of interdiffusion is remarkable because the synthesized invar alloy shows a grain-level uniform distribution of Fe and Ni (Fig. 2i), and Fig. 4d also underpins the chemical homogeneity across a typical sintering neck. Although Ni may also re-dissolve into the FeOx phase34 (flux J3), atom probe tomography results (Extended Data Fig. 7) confirm the negligible role of this process. Under the microstructural state shown in Fig. 4b, when the FeOx reduction initiates, the freshly formed Fe tends to dissolve into the Ni-rich neck and the eminent local concentration gradient further drives interdiffusion, enabling substitutional alloying (Fig. 3e, right). We note that this process may be accomplished through a transient Fe-rich reaction front (several nm thick) at the FeOx/Ni-rich neck interface, without bulk Fe nucleation. This might be responsible for the absence of any metallic Fe phase (bcc) throughout the SXRD diffractograms (Fig. 3b) and detailed atomic-level characterization is advised as future work.
The densification contribution through interdiffusion is also prominent because it naturally facilitates mass transport to the sintering neck35,36,37 and is confirmed to be more effective than Ni self-diffusion (Extended Data Fig. 6). Upon densification, open pores annihilate, resisting the reduction of FeOx because of the retardation of effective H2 transport and the release of H2O (refs. 38,39). This thermodynamic–kinetic jointly governed process implies the crucial role of heating rate in the synthesis: as compared in Fig. 4e, the specimen obtained using a faster heating rate (20 °C min−1) contains considerable residual FeOx phase with reduced porosity, whereas successful synthesis of the invar alloy is achieved with a slower heating rate (5 °C min−1; Extended Data Figs. 3 and 8). This distinction can be explained by the temperature dependency of the fluxes for interdiffusion (J2) and FeOx reduction (J1), as shown in Fig. 4f, in which a crossover point is expected at temperature Ts (see legend of Fig. 4 for semi-quantitative rationalization). Below this temperature, interdiffusion-driven densification is modest compared with the eminent FeOx reduction. With a sufficiently slow heating rate (βi), complete conversion can be attained (Fig. 4f, bottom), leaving excessive porosity in the microstructure (see also Fig. 1c). Conversely, densification becomes more predominant above Ts, resisting the inherently sluggish FeOx reduction, particularly when conversion remains incomplete before Ts under a fast heating rate (βj). These theoretical analyses are supported by experimental observations, in which an evident trade-off is observed between porosity and residual oxide content with respect to heating rate (Fig. 4g). With the foregoing considerations, we also anticipate a complete conversion window bounded by the minimum temperature to activate FeOx reduction and the complete conversion temperature corresponds to the fastest possible heating rate (βc). Within such a temperature window, a tunable design of porosity and grain size is possible (see also Fig. 1c), diversifying the microstructure design space for the ‘one step oxides to alloy’ synthesis approach (Fig. 1a).
The thermodynamic–kinetic insights mentioned above not only theorize the successful synthesis of bulk Fe–Ni invar alloys from oxides, used here as a demonstrator example, but open a new general paradigm of fabricating metallic alloys directly from oxides through a one-step solid-state process (Fig. 1). To demonstrate the universality of this synthesis route, we exemplify in Extended Data Fig. 9 a one-step synthesis also of a ternary fine-grain bulk Fe63Ni32Co5 super invar alloy40 directly from oxides. To generalize our synthesis method to industrial practice, three core factors require consideration. First, effective removal of gangue oxides from natural ores through mechanical separation and hydrometallurgical purification. Second, balancing H2 partial pressure and process temperature. To alleviate the high H2 partial pressure (>0.75) required for complete reduction at 700 °C, improving gas convection in the countercurrent flow furnace or slightly increasing the process temperature up to 800–900 °C is recommended for large-scale production. Third, complete pore annihilation. Although a 0.5-h pressure-free sintering step can reduce the porosity level below 1% for laboratory-scale specimens (Fig. 2k), industrial-scale production might necessitate overlaid hot isostatic pressing to improve the structural integrity of the final product. Finally, for a rough estimation (Extended Data Fig. 10), our ‘one step oxides to bulk alloy’ operation may reduce around 41% energy cost (about 6.97 GJ tonne−1) compared with the traditional multi-step alloy-making approach.
In summary, we report here a redox-inspired sustainable alloy design concept fulfilling one-step synthesis of bulk alloys directly from oxides. Following the thermodynamic guideline and the integrated kinetic conception, we applied this approach to the fabrication of bulk Fe–Ni invar alloys with microstructure–bulk property combinations that are ready to be deployed in real-world applications. The as-synthesized alloy not only exhibits a near-zero thermal expansion property aligning well with the invar alloys fabricated using the traditional multi-step metal extraction, liquid alloying and thermomechanical processing routes but is also accessible to wide microstructure tunability. The universality of our approach, however, goes beyond the specific scope of Fe–Ni binary invar alloy synthesis: the same concept can be extended (1) to various dilute oxide-bonded transition metals and (2) to even highly contaminated oxidized feedstocks of diverse origins. This approach also dissolves some of the classical boundaries between extractive and physical metallurgy, inspiring direct conversion from oxides to application-worthy products in one single solid-state operation.